Regulatory: Software Development for regulated sectors
Software teams, V&V teams, and regulatory software consultancy
We offer expert software development teams and consultants for regulated industries, such as medical, aerospace, automotive, and industrial safety-critical systems. Our teams work closely with you to deliver the highest quality software that meets regulatory standards.
Quality first
High-quality software is vital for sectors that make safety-critical products. It reduces risks and can improve product success and user satisfaction.
Our software development teams use techniques that improve quality and create fast learning cycles. This also brings more flexibility to a project when your priorities change, reassuring you that you are building the right thing.
Regulatory software development teams
With extensive experience working in regulated sectors, we can help at all points of the product lifecycle, from new projects to legacy software. If you have existing internal development teams and processes, our teams can integrate with them.
Our experience includes working on Software as a Medical Device (SaMD) products such as anticoagulation clinical decision support software and AI-based sample diagnostics, as well as medical devices such as a glucose meter, IVF modular workbench, and a therapeutic product for phantom limb pain. We’ve also used our Lean-Agile approach to help products, such as this diabetes self-monitoring device, get FDA compliance.
Quality development activities
Developers follow coding practices that improve quality, such as test-first development, pair programming, and completion checklists. For medical products, we follow the guidance of TIR45.
Early quality verification activities
Testing can occur regularly, and finding defects quicker reduces costs and risks. All our development teams include experienced testers who perform comprehensive testing to confirm that the product works as specified.
User-centred design (UCD)
Human factors can have a significant influence on the success of a product. We have UCD expertise and can support you in meeting the needs of your users. Usability testing of early versions can reveal more insights into what users prefer or need.
Simplified regulatory documentation
Using living documentation to automate documentation reduces effort and ensures the code and documentation are in sync. Making incremental updates to documentation throughout development reduces waste.
Risk management
Risk management activities identify and mitigate risks sooner. We follow ISO 14971 for medical software risk management.
With quality being so crucial to regulated products, Lean-Agile development can give you the edge. It creates regular versions of working software and drives continuous improvement. With over 12 years of Agile experience, we are confident that it gives excellent results and improves customer outcomes. However, we can work with you even if you use Waterfall.
For medical device software or software as a medical device (SaMD), we follow IEC 62304, IEC 82304, and TIR-45. Read more about our medical software experience.
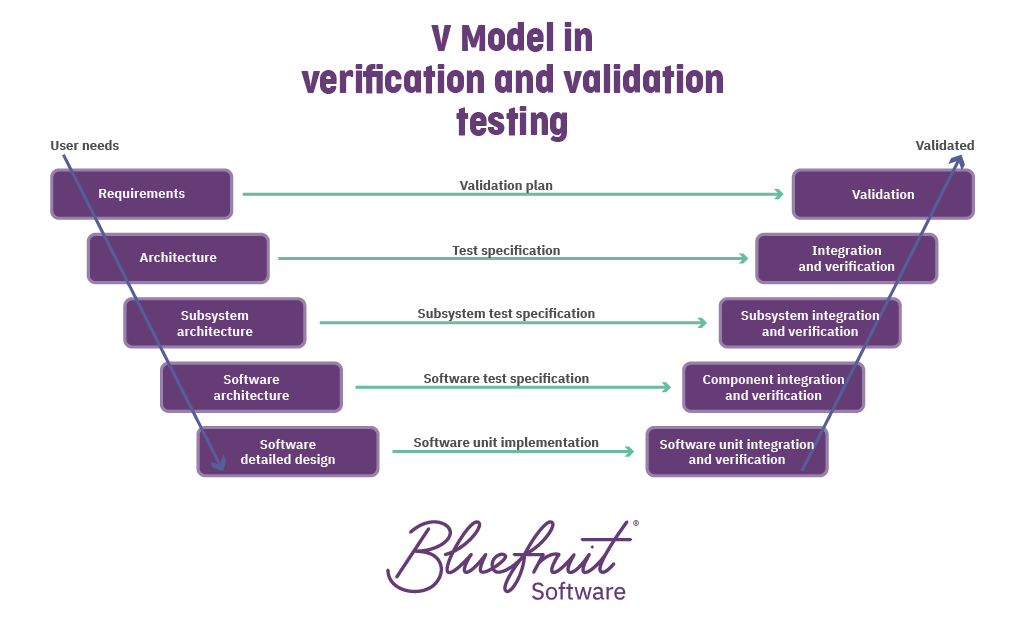
Verification and Validation teams
Verification and validation (V&V) of software requirements is essential to getting FDA or EU/CE approval. Our bespoke V&V teams include quality analysts and testers skilled in verification analysis and regulatory testing for medical devices.
We can work alongside your own product development teams and support you in understanding and meeting the relevant standards. Thinking about V&V during development gives the benefits of fast feedback loops that improve specifications and processes and reduces the time V&V will take at the end of a project. Alternatively, we can help with already completed products.
Whereas development teams might focus on a single feature or area, V&V teams become experts in your project requirements and constantly reflect on the broader picture. This knowledge has helped us drive improvements to your requirements, processes, software, and hardware.
V&V activities can include:
- Testing gap analysis to identify gaps in existing testing.
- Verification test writing and verification testing, ensuring a complete test suite.
- Executable specification writing.
- Manual and exploratory testing.
- Test automation.
- Documentation support and review. Including living documentation setup, requirement review, and compliant software development processes.
- Traceability between requirements and tests.
- Rigorous peer review process.
- Thorough test quality and test result review processes.
- Risk management activities.
- Audit trail of activities (to IEC 62304 requirements).
Regulatory software consultancy
Our consultants have experience working in regulated fields. Our consultancy services include the following:
- IEC 62304 gap analysis – an assessment to identify gaps between your solution and the standard for medical device software – software lifecycle processes.
- IEC 61508 gap analysis — an assessment to identify gaps between your solution and the functional safety standard for industrial control systems and safety-critical software
- Software Quality Audit – an assessment to evaluate the quality of your software solution.
- Compliance support – consulting services for standards and regulatory software development techniques.
- Risk management, including help with ISO 14971.
- Agile coaching and training.
- Living documentation coaching and training.
Compliance with standards
Our software development teams can align with the standards required for your sector.
Medical: We have worked with classes A to C on previous medical device software and SamD projects, using IEC 62304 and FDA requirements 21 CFR Part 11, 21 CFR Part 820, and CE requirements. We can also work with ISO 13485, ISO 14971, and IEC 62366.
Aerospace: Our aerospace experience includes projects that meet the DO-178B standard.
Industrial Systems: For industrial systems, we also develop to IEC 61508, the functional safety standard for safety-critical embedded software. This includes support across the full process — from gap analysis and SIL determination through to compliant development and documentation. See how we helped Apollo Fire Detectors achieve IEC 61508 compliance, or download our IEC 61508 for embedded software teams.
Case studies
Insights from our blog
Are you looking to develop a new product or have an existing one?
Bluefruit Software has been providing high-quality embedded software development and testing services for more than 20 years. Our teams of experienced engineers, testers, and analysts have worked with a diverse range of clients and industries, including medical, industrial, scientific instruments, aerospace, automotive, consumer, and more.
We can help you with software development at any project stage, ensuring quality, reliability, and security. Contact us today to discuss your software development needs.